Emerging contaminants, particularly antibiotics and persistent organic pollutants, pose significant threats to global water security and public health. Researchers from Sichuan University and their collaborators have published a groundbreaking study in the journal Environmental Science and Ecotechnology, revealing how phenolic compounds can enhance the degradation of antibiotics in complex water systems. This research, available online since February 27, 2026, challenges existing notions about pollutant interactions and their effects on water treatment efficiency.
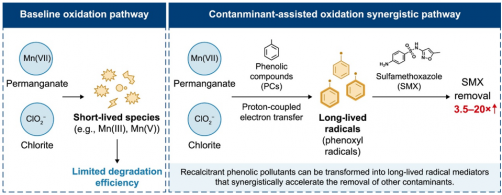
The study focused on how phenolic contaminants impact the removal of antibiotics, specifically using sulfamethoxazole as a model. Traditional advanced oxidation processes often struggle due to the competition among multiple contaminants for reactive intermediates. While many pollutants inhibit the degradation of antibiotics, the findings revealed that phenolic compounds significantly increased sulfamethoxazole degradation rates from around 15% to nearly complete removal within minutes under optimized conditions.
The researchers employed a combination of experimental chemistry, spectroscopy, and theoretical modeling to uncover a new contaminant-assisted oxidation mechanism. They discovered that the enhancement in antibiotic degradation was not due to conventional reactive oxygen species. Instead, phenolic molecules engaged in proton-coupled electron transfer reactions with permanganate and chlorite, resulting in the formation of stable phenoxyl radicals. These radicals proved to be long-lived, allowing them to continue the degradation of antibiotics even after the initial reaction had occurred.
Redefining Treatment Strategies
Advanced spectroscopic trapping experiments confirmed the presence of phenoxyl radicals, while inhibition tests indicated that their removal halted antibiotic degradation entirely. Computational modeling further illustrated that hydrogen-bond-mediated electron transfer was responsible for the formation of these radicals, revealing that only certain phenolic structures had the capability to generate significant acceleration effects.
The study also highlighted the selective behavior of these radicals, which preferentially attacked amino-containing antibiotics through electron transfer followed by radical–radical coupling reactions. This selectivity correlated with the hydrophobicity of the pollutants, suggesting a novel mechanism that contrasts with what is typically observed in inorganic oxidation systems. Remarkably, the radicals maintained their effectiveness in real water matrices containing inorganic ions and natural organic matter, demonstrating resilience to environmental interferences.
The findings prompt a reevaluation of the traditional view that the coexistence of contaminants is detrimental to water treatment. By showing that phenolic pollutants can act as reactive mediators, the research advocates for a shift in perspective from eliminating these compounds to engineering beneficial interactions that enhance treatment performance.
The researchers emphasized that the long-lived phenoxyl radicals exhibit stability, selectivity, and a tolerance to matrix variability—qualities that are rarely achieved concurrently in advanced oxidation systems. This insight could lead to the development of adaptive remediation strategies that handle increasingly complex wastewater compositions.
Implications for Wastewater Treatment
The study suggests innovative opportunities for treating pharmaceutical wastewater, where phenolic byproducts and antibiotics often coexist. Instead of preemptively removing phenolic compounds, treatment systems could harness them to improve oxidation efficiency through controlled pre-oxidation stages. Such strategies could enhance pollutant removal while simultaneously reducing chemical consumption and operational costs.
The research aligns with a broader movement toward “self-adaptive” remediation technologies that leverage contaminant networks instead of treating pollutants in isolation. Future work will focus on pilot-scale testing, process optimization, and intelligent control systems capable of adjusting oxidant dosing according to fluctuating wastewater conditions.
Ultimately, this study points toward smarter water treatment designs that turn the complexity of pollution into a functional advantage, paving the way for more effective management of water resources and public health. The authors express gratitude to funding sources, including the National Key Research and Development Program of China, National Natural Science Foundation of China, and the Sichuan Science and Technology Program, for their support in this research initiative.