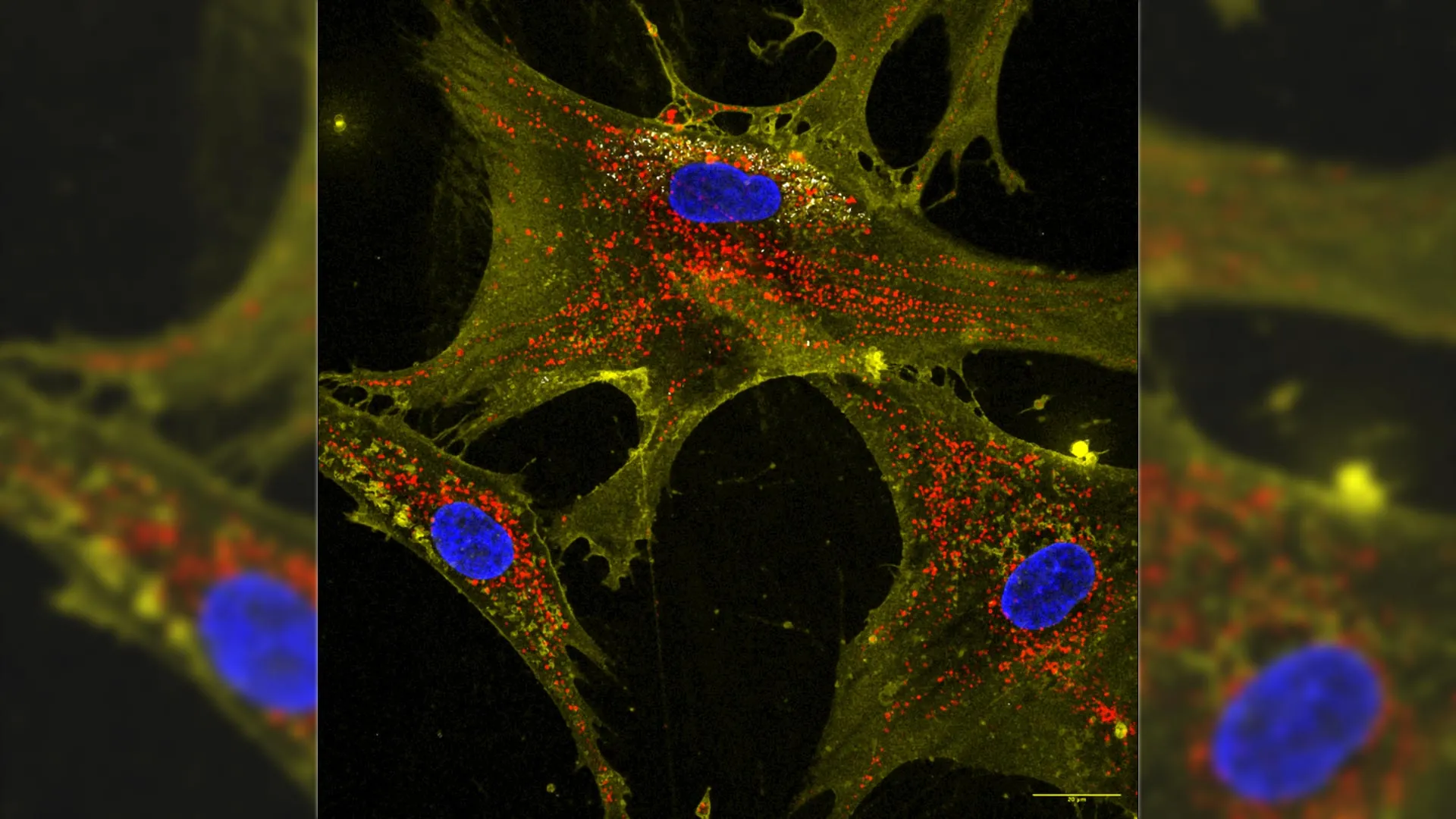
Biomedical researchers at Texas A&M University have developed a groundbreaking technique that enhances the ability of stem cells to produce mitochondria, the energy-producing structures within cells. This innovative method, which utilizes microscopic particles known as nanoflowers, could potentially revolutionize treatments for aging tissues and various degenerative diseases.
The research, published on November 27, 2025, in the *Proceedings of the National Academy of Sciences*, demonstrates that stem cells exposed to these nanoflowers can produce up to double the normal quantity of mitochondria. When these energized stem cells are placed alongside damaged or aging cells, they transfer their surplus mitochondria, effectively rejuvenating the energy production of the impaired cells.
Dr. Akhilesh K. Gaharwar, a professor of biomedical engineering at Texas A&M, emphasized the significance of their findings, stating, “We have trained healthy cells to share their spare batteries with weaker ones.” This process improves the vitality of aging or damaged cells without the need for genetic modifications or pharmaceuticals.
The decline in mitochondrial function is associated with aging and various health conditions, including heart disease and neurodegenerative disorders such as Alzheimer’s disease. As cells age or suffer damage, their mitochondrial count decreases, leading to reduced energy production and overall cell health. The innovative approach taken by the Texas A&M team could address this issue by enhancing the body’s natural ability to replenish worn-out mitochondria.
Enhancing Mitochondrial Production
The nanoflowers are composed of molybdenum disulfide, an inorganic compound capable of forming diverse two-dimensional shapes at nanoscale. By incorporating these nanoflowers with stem cells, the researchers have created what they describe as “mitochondrial biofactories.” These biofactories can transfer two to four times more mitochondria to neighboring damaged cells than untreated stem cells.
Lead author John Soukar remarked on the efficiency of this method, comparing it to giving an old electronic device a new battery pack. “Instead of tossing them out, we are plugging fully-charged batteries from healthy cells into diseased ones,” he explained. The results indicate that the revitalized cells not only exhibit enhanced energy levels but also display increased resistance to cell death, even when subjected to harmful treatments like chemotherapy.
Previous methods aimed at increasing mitochondrial production often require frequent dosing and are less effective in the long term. In contrast, the larger nanoparticles used in this study remain within the cells and continue to stimulate mitochondrial production more persistently. This could mean that therapies based on this nanoflower technology may only need to be administered around once a month.
Potential Applications Across Medical Fields
The flexibility of this technique presents promising possibilities for treating various conditions. While the research is still in its early stages, its application could extend to different tissues throughout the body. According to Soukar, “You could put the cells anywhere in the patient. For cardiomyopathy, you can treat cardiac cells directly, or if you have muscular dystrophy, you can inject them right into the muscle.”
The project has received funding from the National Institutes of Health, the Welch Foundation, the Department of Defense, and the Cancer Prevention and Research Institute of Texas. Additional support has come from the President’s Excellence Fund at Texas A&M University and the Texas A&M Health Science Center Seedling Grant. Key collaborators included fellow Texas A&M researchers Dr. Irtisha Singh, Dr. Vishal Gohil, and Dr. Feng Zhao.
As this research progresses, it holds the potential not only to enhance stem cell therapies but also to pave the way for innovative treatments for a range of age-related and degenerative diseases. If future studies confirm these encouraging results, the implications for medical science could be profound, opening new avenues for combating the effects of aging and cellular damage.