Researchers at Beihang University have developed a new material that efficiently removes copper from water, achieving a remarkable 99.5% removal rate. The innovative solution, known as ferromanganese oxide-modified biochar (FMBC-600), also reduces total organic carbon by 92.6%. This advancement addresses the growing issue of freshwater scarcity and the need for effective wastewater treatment methods.
Traditional treatment approaches often fail to tackle stable metal complexes, particularly those bound with organic agents such as citric acid. These complexes, frequently found in industrial effluents from sectors like electroplating and textile dyeing, present significant ecological and health risks due to their resistance to degradation. The new biochar material offers a promising alternative, designed to overcome the limitations of conventional treatments which primarily target free metal ions.
Developed by a team led by Wenhong Fan, the research findings were published in Biochar X on October 14, 2025. The study details the synthesis of FMBC-600 through a process involving impregnation and high-temperature calcination. The resultant biochar features a rough surface uniformly coated with nanoparticles measuring 80–100 nm, which enhances its adsorption capabilities.
Innovative Adsorption Mechanisms
The modified biochar employs a dual adsorption mechanism. It combines chemical bonding facilitated by oxygen-containing functional groups with physical adsorption enabled by its porous structure. Advanced characterizations, including FE-SEM and EDS analyses, confirmed the successful incorporation of iron (Fe) and manganese (Mn) into the biochar matrix, as well as subsequent copper deposition after treatment.
Further analysis through FTIR and XRD indicated the presence of functional groups such as hydroxyl and aromatic structures, alongside crystalline phases of Mn3O4 and (FeO)0.099(MnO)0.901. XPS results highlighted the roles of Fe and Mn in the adsorption process, suggesting electron transfer and surface complex formation as key mechanisms.
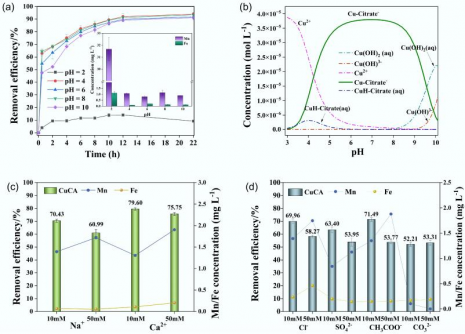
The researchers optimized the production process, identifying an ideal iron-to-manganese ratio of 1:4, a manganese concentration of 0.03 M, and pyrolysis at 600 °C. Under these conditions, FMBC-600 demonstrated its ability to remove copper efficiently within 30 minutes, maintaining stability across a pH range of 4 to 10. Notably, even in the presence of competing ions such as sodium (Na+), calcium (Ca2+), chloride (Cl−), and sulfate (SO42−), the material retained high performance, underscoring its selectivity and robustness.
Potential Applications and Future Impact
The findings indicate that FMBC-600 not only excels in removing copper but also showcases excellent stability and reusability, with about 80% efficiency retained after two cycles. This positions the material as a cost-effective solution for large-scale wastewater treatment, particularly in industries that generate persistent heavy metal complexes.
Beyond wastewater applications, the technology has implications for soil remediation, potentially reducing heavy metal accumulation in agricultural lands. Its adaptability to varying water chemistry makes it a practical choice for real-world systems.
The development of FMBC-600 represents a significant advancement in sustainable water treatment technologies. With a straightforward and low-cost production process, it offers a feasible option for managing industrial effluents and contributes to global efforts to achieve clean water and sustainability goals.
This research was supported by the National Natural Science Foundation of China and various regional educational projects, reflecting a commitment to innovative solutions in environmental sustainability. The comprehensive results can be located in detail in the publication available at DOI: 10.48130/bchax-0025-0001.