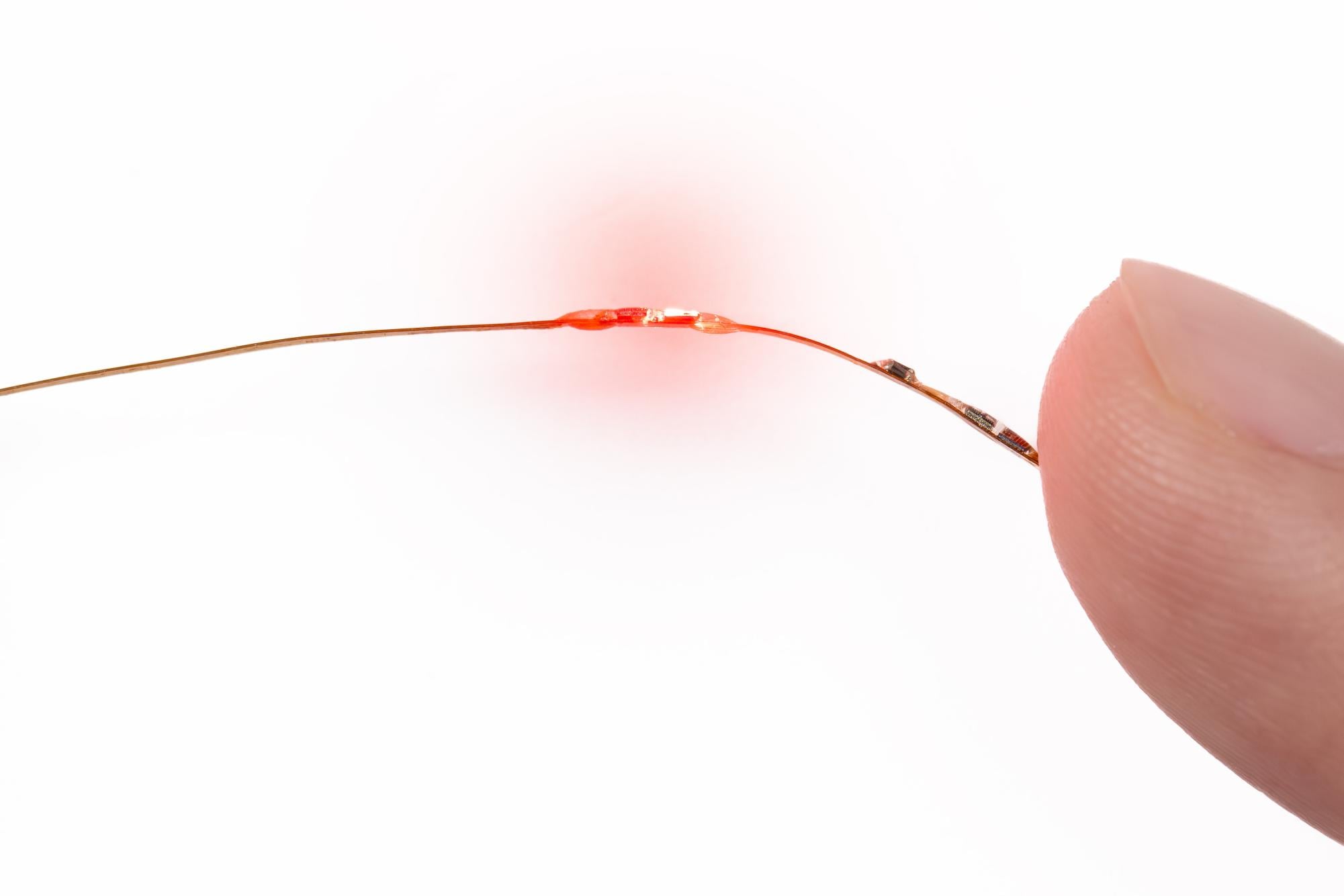
Researchers at Northwestern University have developed a groundbreaking hair-thin device capable of continuously monitoring vital signs in fetuses during surgical procedures. This innovative technology, created in collaboration with Ann & Robert H. Lurie Children’s Hospital in Chicago, represents a significant advancement in fetal surgery, enabling medical professionals to track critical health metrics throughout operations.
A study published on January 26, 2023, in the journal Nature Biomedical Engineering outlined the device’s capabilities. The tool measures essential parameters such as heart rate, temperature, blood oxygen levels, and heart rate variability through small ports in the body specifically designed for fetal surgery. According to John Rogers, a professor at Northwestern and director of the Querrey Simpson Institute for Bioelectronics, current methods do not allow for continuous monitoring of these vital signs in fetuses.
Rogers emphasized the necessity of real-time data, stating, “You’d like to be able to track the patient’s status throughout the surgery, so that you could detect any kind of adverse effect as soon as possible, so that the surgeon could take corrective action.” He noted that the device employs “extremely miniaturized” sensors and uses small balloons to navigate through the ports while ensuring the appropriate level of contact with the fetus.
The development of this device comes at a crucial time, as fetal surgeries, while uncommon, can be vital in preventing potential complications after birth. Steven Papastefan, a general surgery resident at Northwestern Medicine and co-author of the study, explained that the current technique for monitoring fetal heart rate during surgery, transuterine echocardiography, is limited. Surgeons can only conduct these checks every ten minutes due to the constraints of the surgical environment.
“What this device allowed us to do is to basically continuously monitor the fetus throughout the operation so that we know on a second-by-second basis how it’s doing,” Papastefan said. He highlighted that many fetal surgeries aim to address conditions that could lead to irreversible damage, such as twin-twin transfusion syndrome and spina bifida.
The impact of this device could be profound. Aimen Shaaban, director of the Chicago Institute for Fetal Health at Lurie Children’s Hospital, explained that spina bifida involves an opening in the spine that can lead to severe neurological problems. Intervening before birth can significantly reduce the risk of complications, allowing for a healthier outcome for the newborn. “The baby can have stronger legs and potentially avoid the severity of hydrocephalus,” Shaaban noted.
The collaboration between Rogers’ and Shaaban’s teams exemplifies a successful partnership in advancing medical technology. Testing of the device involved surgical procedures on lambs, where researchers simulated spina bifida, followed by the use of the device during repair operations. While the device has yet to be tested on humans, Rogers anticipates that initial human trials could begin two to three years after obtaining regulatory approval from the FDA.
At Lurie Children’s Hospital, fetal surgeries occur approximately once or twice each week. Shaaban expressed optimism about the device’s potential, stating, “It’s really going to change the way we actually care for the patients and give us just a greater insight into what we’re actually doing.” The introduction of continuous monitoring could enhance surgical outcomes and provide surgeons with critical data to make informed decisions during operations.
This innovative development marks a significant step forward in fetal medicine, promising to improve surgical interventions and outcomes for vulnerable patients.