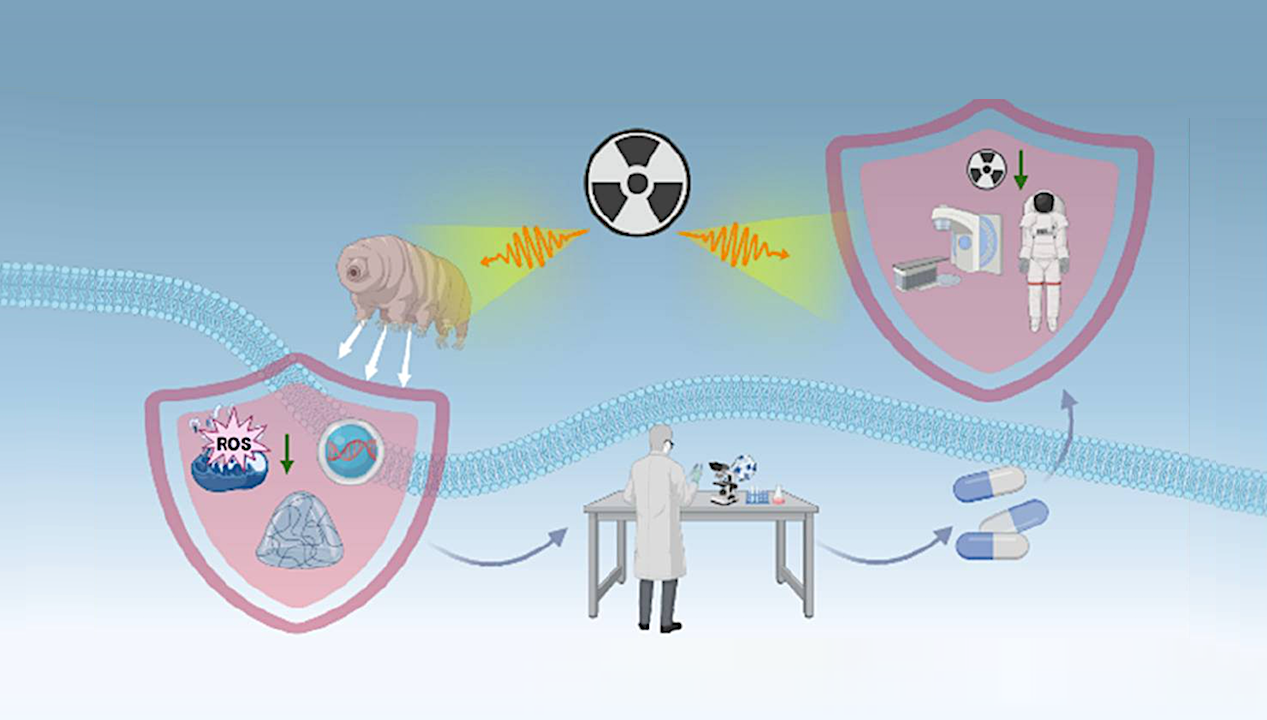
Recent advancements in drug discovery are drawing inspiration from tardigrades, tiny organisms known for their remarkable resilience to extreme conditions, including high levels of ionizing radiation. Researchers are exploring how the unique biological features of tardigrades can lead to innovative strategies for protecting human health against radiation exposure, particularly in medical and space exploration contexts.
Ionizing radiation poses serious health risks, causing double-strand DNA breaks and oxidative stress. These effects are the driving factors behind conditions such as acute radiation syndrome, secondary malignancies, and significant toxicity during radiotherapy. Conventional radioprotectants—such as aminothiols, antioxidants, and cytokines—often offer limited protection, as their effectiveness is curtailed by narrow therapeutic windows and systemic toxicity.
In stark contrast, tardigrades, classified under the phylum Tardigrada, have evolved a robust defense mechanism, allowing them to withstand radiation doses exceeding 5 kGy. This extraordinary capability is attributed to several key adaptations, including:
1. **Genome-shielding proteins**: Notably, the damage suppressor (Dsup) and Tardigrade DNA-Repair protein 1 (TDR1) provide a protective coating for chromatin, reducing DNA damage.
2. **Intrinsically disordered proteins**: These proteins help to vitrify cellular components, limiting the diffusion of damaging radicals.
3. **Antioxidant pigments**: Acquired through horizontal gene transfer, these pigments neutralize reactive oxygen species, further enhancing cellular protection.
4. **Efficient DNA repair systems**: Tardigrades possess hyper-efficient mechanisms for repairing DNA damage and restoring genomic integrity after exposure to radiation.
Exploring the intersection of tardigrade biology and translational pharmacology opens new avenues for emerging radioprotective therapies. Researchers are investigating various modalities, including recombinant proteins, cell-penetrating proteins, mRNA therapeutics, and biomimetic nanomaterials. Each of these approaches seeks to enhance the protective qualities of traditional treatments while addressing critical challenges such as bioprocess scalability, macromolecule stability, immunogenicity, and targeted delivery.
The insights gained from studying these extremophiles suggest that tardigrade-inspired interventions could significantly improve patient safety during cancer treatments, reduce casualties in nuclear incidents, and protect astronauts on deep-space missions. By transforming an evolutionary curiosity into a powerful set of medical countermeasures, the field of radioprotection is poised for a substantial transformation.
The potential applications of this research are vast. For instance, in the context of cancer therapy, improved radioprotectants could help shield healthy tissues from the harmful effects of radiation, thereby enhancing treatment efficacy and reducing side effects. Additionally, the ability to safeguard against radiation exposure in nuclear accidents could lead to better preparedness and response strategies, potentially saving lives.
As the understanding of tardigrade biology expands, the implications for medicine and space exploration become increasingly promising. Through continued collaboration between biologists and pharmacologists, the vision of a future where human health can be safeguarded against radiation may soon become a reality.