A team from the University of Minnesota has developed a groundbreaking one-pot method for synthesizing blue light-responsive aryne precursors from commercially available carboxylic acids. This innovative approach simplifies the previously complex process of generating these valuable organic intermediates, opening new avenues for drug discovery and agricultural chemistry. The findings were published on November 11, 2025, in the journal Nature.
Arynes are highly reactive organic compounds that contain a triple bond within an aromatic ring. Their unique chemical structure allows them to engage with a wide array of functional groups, making them essential in the development of complex aromatic molecules. Despite their potential, synthetic chemists have often avoided using arynes due to the challenges in their synthesis.
For many years, traditional methods to create arynes involved harsh bases that strip protons from strong C–H bonds, followed by halide elimination. These techniques can be unsuitable for sensitive molecules, leading to a limited application of arynes in organic reactions. Alternative methods, such as thermally activated precursors, have been found to be highly unstable, while UV-light approaches often result in unwanted side reactions.
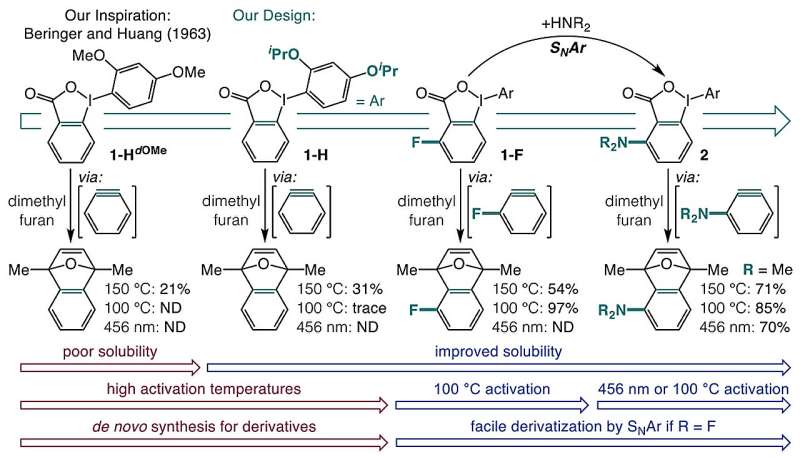
The research team sought to address these limitations by exploring the use of o-iodoniobenzoates as aryne precursors that could be activated by blue light or mild heat. Through their investigation, they devised a one-pot synthesis method that transforms carboxylic acids into these precursors. Initially, they faced challenges due to the poor solubility of o-iodoniobenzoates and the tendency for unwanted side reactions.
After extensive experimentation, the researchers discovered that introducing isopropoxy groups significantly improved solubility and minimized undesired reactions. Furthermore, they found that adding a substituent adjacent to the carboxylate group allowed for the generation of aryne precursors that could be activated by blue light or heating up to 100 °C.
Their investigations revealed that heat activation is primarily driven by a field effect, where chemical groups near the carboxylate on the aromatic ring enhance decarboxylation. Conversely, activation through blue light (at a wavelength of 398 nm) excites the molecule into a triplet state. This excitation causes the aromatic ring to detach from the iodine, facilitating the loss of carbon dioxide and leading to the formation of arynes.
The implications of this new synthesis method are significant. Researchers highlight that it unlocks access to a diverse range of aryne precursors starting from common carboxylic acids. This method is compatible with various functional groups, greatly simplifying the synthesis of complex aromatic compounds. As a result, it paves the way for advancing pharmaceutical and agrochemical research, potentially revealing previously unexplored chemical territories.
This study, led by Chris M. Seong and his team, represents a notable advancement in synthetic chemistry. The team’s findings offer a practical and efficient approach to generating aryne derivatives, making them more accessible for a broader range of applications in organic synthesis.